When planning a bone graft, in addition to the characteristics regarding the patient and defect to be repaired, a number of details must be considered by the dental surgeon, especially those associated with the biomaterial itself, such as: morphology/particle size, porosity and surface morphology, as such factors will guide how the host will interact with the graft immediately after the implant.1
Therefore, POROSITY must be one of the priorities to be observed, as it is known that the kinetics of osseointegration is related to the way a bone tissue develops three-dimensionally between and within the macrogeometry of biomaterials.1
A common mistake is to believe that porosity is exclusively linked to the origin of the biomaterial when other factors also influence this characteristic. For example, materials of the same composition may have different morphologies and porosities, due to their production process.
Below, through electron microscopy images, we can compare the porosity of different biomaterials:
Foreign Biomaterial x Nanosynt

Images from electron microscopy show that Nanosynt (B³ and D⁵) has greater porosity when compared to the leading bovine biomaterial in the market (A² and C⁴). Images at a magnification of 1,400x (A²) x 1,500 (B³) and 10,000x (C⁴ and D⁵).
Note Nanosynt’s ultra-porous surface in relation to bovine. This condition will allow greater permeability and cell anchorage, favoring tissue repair efficiency.
When compared to other synthetic biomaterials, although they are chemically identical (60% hydroxyapatite and 40% B-TCP), Nanosynt’s ultra porous structure contrasts sharply to the marbled pattern of the global-benchmark competitor. Greater permeability favors not only usability, but also the speed of tissue repair.
Competing synthetic biomaterial x Nanosynt
As seen in the images, among the best-known biomaterials on the market, Nanosynt is the only one with such porosity.

Nanosynt biomaterial approach at 30,000xNanosynt biomaterial approach at 30,000x.
This unique feature makes it exceptionally hydrophilic and suitable for cell anchorage. Its interconnected structure is valuably well assimilated by the body during the tissue remodeling process, enabling unique cell interaction.

This favorable potential for bone growth is clinically evidenced and has already been published in a reputable international journal:
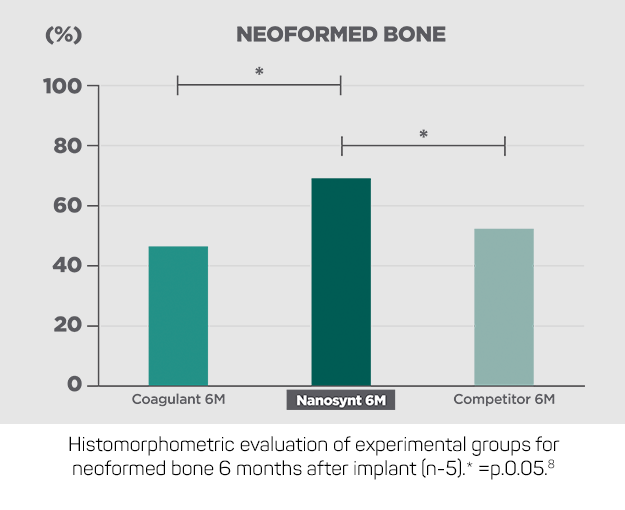
As can be observed, the interconnected structure, high porosity and biphasic chemical composition make Nanosynt gain 20% more neoformed bone in a six -month period when compared to its main competitor.
Consolidation of synthetic materials as bone substitutes is a scientific reality. Nanosynt, for example, reaches temperatures above 1000°C in its production process, making the chances of contamination null. In addition, its industrial process follows international purity standards, ensuring a final product that safely meets the highest quality standards.
Notwithstanding, benefits associated with usability, quick handling, fractional portions and excellent cost-benefit make Nanosynt the best option when choosing your biomaterial.
References
- Tovar N, Jimbo R, Witek L, et al. The physicochemical characterization and in vivo response of micro/nanoporous bioactive ceramic particulate bone graft materials. Mater Sci Eng C 2014; 43: 472–480.
- Lambert F, Bacevic M, Layrolle P, et al. Impact of biomaterial microtopography on bone regeneration: comparison of three hydroxyapatites. Clin Oral Implants Res 2017; 28: e201–e207.
- Oliveira LA de. Imagens internas – Nanosynt.
- Desterro F de P do, Sader MS, Soares GD de A, et al. Can inorganic bovine bone grafts present distinct properties? Braz Dent J 2014; 25: 282–288.
- Perdigão J. Imagens internas – Nanosynt.
- Dietze S, Bayerlein T, Proff P, et al. The ultrastructure and processing properties of Straumann Bone Ceramic® and Nanobone®. Folia Morphol (Warsz) 2006; 65: 63–65.
- Coutinho V. Avaliação do efeito da radiação laser em cultura de células cultivadas sobre um substrato sintético nanoparticulado: caracterização química, morfológica e análise de sua biocompatibilidade tecidual in vivo. Universidade Cruzeiro do Sul, 2017.
- Uzeda MJ, de Brito Resende RF, Sartoretto SC, et al. Randomized clinical trial for the biological evaluation of two nanostructured biphasic calcium phosphate biomaterials as a bone substitute. Clin Implant Dent Relat Res 2017; 19: 802–811.