Authors: Eng. M.Sc. William de Souza Wiggers e DDS M.Sc Rafael Cury Cecato
The emergence of implantology at the beginning of the 20th century, with Payne and Greenfield, the forerunners of the specialty, presented solutions such as gold and porcelain. Payne described the implant of a cylindrical gold basket, placed after enlarging the diameter of the alveolus with the aid of a drill. Since then, a lot of technology and innovation have been implemented, many products on the market have been improved and new techniques have emerged to contribute to the high success rate of endosteal implants.
The literature presents scientific references that prove the efficiency of solutions in this area and one of the highlights is the frictional conical connection, which provided Odontology with new planning and execution possibilities, which brought to implantology professionals the ability to deliver better results in esthetics, function and peri-implant health, with more efficiency in rehabilitation on dental implants.
“The exclusive Arcsys Implant System confirms the FGM brand’s evolution, anticipating market needs with all its folding possibilities. The line has CE certification, a seal that confirms the quality of the products in accordance with the European Community’s regulations and the differentials are due to the expressive reduction in the volume of components in stock, more refined planning, with greater predictability in the results, one of the highlights being the bacterial seal present in the connection of this system.” Rafael Cury Cecato – Senior scientific consultant (Biomaterials)

Absolute and relative frequencies of media with bacterial growth, after 48 hours, in different prosthetic component angles, cycled and non-cycled⁵.
LITERATURE SHOWS THAT INTERNAL CONICAL CONNECTIONS ARE MORE EFFICIENT IN DISSIPATION OF FORCES GENERATED IN THE INTERFACE BETWEEN IMPLANT AND COMPONENT, AS WELL AS IN
PROSTHETIC ABUTMENT STABILITY¹⁻⁴.
In a study carried out by the researchers associated with Prof. Dr. Elizabeth Martinez⁵, from Faculdade São Leopoldo Mandic, evaluating precisely the microbiological sealing on Arcsys frictional implants, it has been proven its effectiveness in 100% of the samples (Table 1)⁵.
These results were confirmed both in the conditions cycled as non-cycled, for the groups with straight prosthetic components and with 5, 10 and 20 degrees, attesting to the efficiency of the Arcsys System in various clinical conditions.
Due to the differential of customizing the prosthetic components’s angle by up to 20 degrees, it is possible to optimize the relationship between positioning the implant and dental prosthesis, without compromising the functional characteristics of the component.
This ability is made possible by the use of a stainless steel alloy, perfectly biologically viable⁶, in the manufacture of the prosthetic component, associated with the use of a device, making it possible to adjust the component’s angle.


1.1 and 1.2 | Cross-sectional and longitudinal images of the connection between implant and Arcsys frictional component, captured by microCT, demonstrating the absence of gaps in the connection
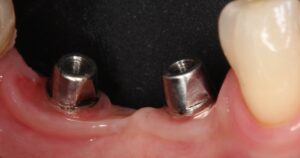
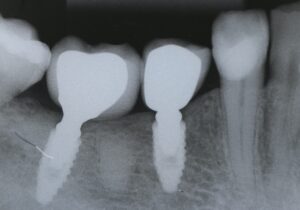
2.1 and 2.2 | Clinical and radiographic appearance of peri-implant tissues after working 5 years
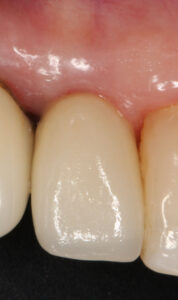
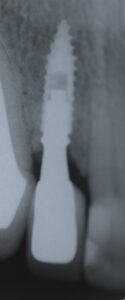
3.1 and 3.2 | Clinical and radiographic appearance of peri-implant tissues after working 4 years
In the previously referenced study, the proven bacterial sealing of the frictional connection of the Arcsys Implant System solves possible future complications related to bacterial percolation at the implant/component interface, removing a possible and important cause of the development of peri-implant diseases.
This adaptation is ratified when analyzing that the frictional connection promotes a “cold weld” between the prosthetic component and the implant, evidenced in the images of scanning electronic microscopy5,7, thus avoiding the formation of spaces in the interface of the set, promoting the sealing and avoiding bacterial colonization of the region. This effective microbiological sealing provides biological, functional and esthetic results superior to those found in other systems available on the market.
BIBLIOGRAPHIC REFERENCES
1. Koutouzis T, Wallet S, Calderon N, Lundgren T. Bacterial Colonization of the Implant–Abutment Interface Using an In Vitro Dynamic Loading Model. J Periodontol 2011; 82: 613–618.
2 Alves DCC, de Carvalho PSP, Elias CN, Vedovatto E, Martinez EF. In vitro analysis of the microbiological sealing of tapered implants after mechanical cycling. Clin Oral Investig 2016; 20: 2437–2445.
3 Alves DCC, de Carvalho PSP, Martinez EF. In vitro microbiological analysis of bacterial seal at the implant-abutment interface using two morse taper implant models. Braz Dent J 2014; 25: 48–53.
4 Bozkaya D, Müftü S. Mechanics of the taper integrated screwed-in (TIS) abutments used in dental implants. J Biomech 2005; 38: 87–97.
5 de Aguiar Vilela Júnior R, Cláudio Aranha L, Nelson Elias C, Ferreira Martinez E. In vitro analysis of prosthetic abutment and angulable frictional implant interface adaptation: mechanical and microbiological study. J Biomech 2021; 128: 110733.
6 Cecato RC, Martinez EF, Benfatti CAM. Analysis of the Viability and Morphology of Gingival Cells on Materials Used in Novel Prosthetic Components: In Vitro Study. J Contemp Dent Pract 2022; 23: 22–30.
7 Costa MB, Ferreira LFACM, Takeshita WM, Trento CL. Scanning electron microscopic evaluation of microgap size in Morse tarer with straight and angled abutments. Gen Dent 2022; 70: 34–39.